
Hydrogen and Helium in Airship Operations. The two primary lifting gases used by airships have been hydrogen and helium. Hydrogen is the earth’s lightest element, and it can be obtained. Army airship Roma in 1. Army hydrogen airship Roma. Norfolk, VA, February 2. Helium’s non- flammable nature makes it the only practical lifting gas for manned lighter- than- air flight, but it is scarce. Lifting Gas. Much of an airship’s weight is fixed (the dead weight of the ship’s structure and engines, and required weight. But since helium has always been a rare and expensive gas, helium airships began their flights only 9. Hydrogen ships simply released inexpensive and easy- to- replace hydrogen, but helium- inflated ships required equipment. Navy in 1. 92. 4 it was inflated with hydrogen, and the ship. When the United States Navy operated the same ship with helium, as U. S. S. 1. 0, from Rio de Janeiro to Friedrichshafen on April 6, 1. Chem. 4Kids. com: Elements & Periodic Table. Periodic Table and the Elements. Now we're getting to the heart and soul of the way the Universe works. You know that a generic atom has some protons and neutrons in the nucleus and some electrons zipping around in orbitals. Appearance and Characteristics. Harmful effects: Silicon is not known to be toxic, but if breathed in as a fine silica/silicate dust it may cause chronic respiratory. History Scientific discoveries. The first evidence of helium was observed on August 18, 1868, as a bright yellow line with a wavelength of 587.49 nanometers in the. Helium is one of the so-called noble gases. Helium gas is an unreactive, colourless, and odourless monoatomic gas. Helium is available in pressurised tanks. Elements and Compounds around us 193 SCIENCE SCIENCE 5.2. The unscrambled words such as iron, copper, gold, oxygen and carbon that we come across. Introduction: Definitions and mutation rates. We have been using the term 'mutation' pretty loosely up to this point in the course.now we need to define it more. The elements in a compound are always present in the same ratio.? It is easy to separate a compound into the elements that it is made from. As we move past the first eighteen elements, you can start to learn about transition elements in the fourth period (row) of the periodic table. The transition metals. There are several patterns in the table above. Common gases at room temperature include both elements (such as H 2 and O 2) and compounds (such as CO 2 and. 
3 Common Compounds Of Helium BalloonWhen those pieces start combining in specific numbers, you can build atoms with recognizable traits. If you have eight protons, neutrons and electrons, you will have an oxygen (O) atom. If you have seven protons, neutrons, and electrons, you will have a nitrogen (N) atom. The atoms for each element are unique, even though they are all made of similar subatomic parts. Everything is made of atoms. The term 'element' is used to describe atoms with specific characteristics.    
3 Common Compounds Of Helium ShortageThere are almost 1. For example, you are made up of billions of billions of atoms but you probably won't find more than 4. Chemists have learned that over 9. H), carbon (C), nitrogen, oxygen, phosphorus (P), and calcium (Ca). Up to this point in time, we have discovered or created about 1. Scientists just confirmed the creation of element 1. 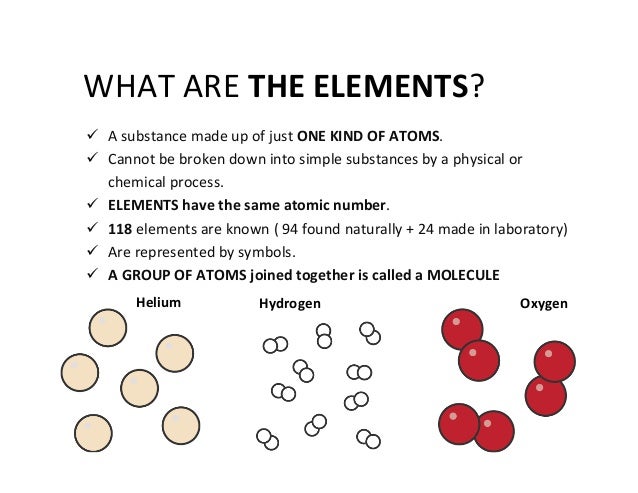
While there are more elements to discover, the basic elements remain the same. Iron (Fe) atoms found on Earth are identical to iron atoms found on meteorites. The iron atoms in the red soil of Mars are also the same. You will never stop discovering new reactions and compounds, but the elements will be the same. Remember that the orbitals are the places you will generally find the electrons as they spin around the nucleus. The transition metals have electron configurations that are a little different from the first eighteen. Make sure you understand the basics of electron orbitals before you move on to this row. Helium»the essentials . Helium gas is an unreactive, colourless, and odourless monoatomic gas. Helium is available in pressurised tanks. Helium is the second most abundant element in the universe after hydrogen. Speaking after breathing an atmosphere rich in helium results in a squeaky voice (don't try it!). Helium is present in the atmosphere at about 0. USA. Its origin in these gases is traced to the decay of radioactive elements in rocks. Emma's first birthday balloon is filled with helium and so rises in air. Helium: historical information. Helium was discovered by Sir William Ramsay and independently by N. Cleve in 1. 89. 5 at London, England and Uppsala, Sweden. Origin of name: from the Greek word . It was not possible to produce this line in the laboratory. Sir Norman Lockyer (1. English astronomer, recognised that no known element at that time gave this line and named the element helium for the sun. For many years helium was regarded as an element that might exist on the sun although it was unknown on the Earth. Spectroscopists at the time doubted the results concerning helium. However the claims initiated a search for the new element on planet earth. In 1. 89. 5, Sir William Ramsay discovered helium after treating cleveite, a uranium mineral, with mineral acids. Ramsey sent samples of the gas to Sir William Crookes and Sir Norman Lockyer who identified helium. It was discovered independently in clevite by Cleve and Langley at about the same time. Lockyer and Professor Edward Frankland suggested the name helium. Helium has no biological role. Helium is present in the atmosphere at about 0. USA. Its origin in these gases is traced to the decay of radioactive elements in rocks. Some minerals contain occluded helium and this can be liberated by heating. Some rocks consist of minerals which contain uranium and potassium. These decay to helium and argon and analysis of these gases can be used to determine the age of the rock. The bulk of the western world's supply is obtained from wells in the USA. Helium is the second most abundant element in the universe. It is very common in the hotter stars. It is an important component in the proton- proton reaction and the carbon cycle in stars. The solid state structure of helium is: ccp (cubic close- packed). Helium: orbital properties Read more »Helium atoms have 2 electrons and the shell structure is 2. The ground state electronic configuration of neutral Helium is 1s. Helium is 1. S0. Isolation. Isolation: there is very little helium on earth as nearly all present during and immediately after the earth's formation has long since been lost as it is so light. Just about all the helium remaining on the planet is the result of radioactive decay. While there is some helium in the atmosphere, currently its isolation from that source by liquefaction and separation of air is not normally economic. This is bacause it is easier, and cheaper, to isolate the gas from certain natural gases. Concentrations of helium in natural gas in the USA are as high as 7% and other good sources include natural gas from some sources in Poland. It is isolable from these gases by liquefaction and separation of from the natural gas. This would not normally be carried out in the laboratory and helium is available commercially in cylinders under pressure. Helium has two isotopes but it consists almost entirely of He- 4 with natural He only containing just over 0. He- 3. Thousands of liters of He- 3 are used annually in cryogenic applications and He- 3 is also used as a neutron counter in nuclear application. He- 3 is also applied in magnetic resonance imaging. Table. Stables isotopes of helium.  
Isotope. Mass/Da. Naturalabund.(atom %)Nuclearspin (I)Nuclearmagneticmoment (.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2017
Categories |
 RSS Feed
RSS Feed
